-
Author
Carlos Maturana -
PI
Thomas J. Kremen, MD
-
Co-Author
Brendan Shi MD, Shannon Wu BS, Brandon Peterson MS, Tyler Clites PhD
-
Title
Reduced Cementation in the Setting of Reverse Shoulder Arthroplasty with Proximal Bone Loss: A Biomechanics Study
-
Program
STTP
-
Other Program (if not listed above)
-
Abstract
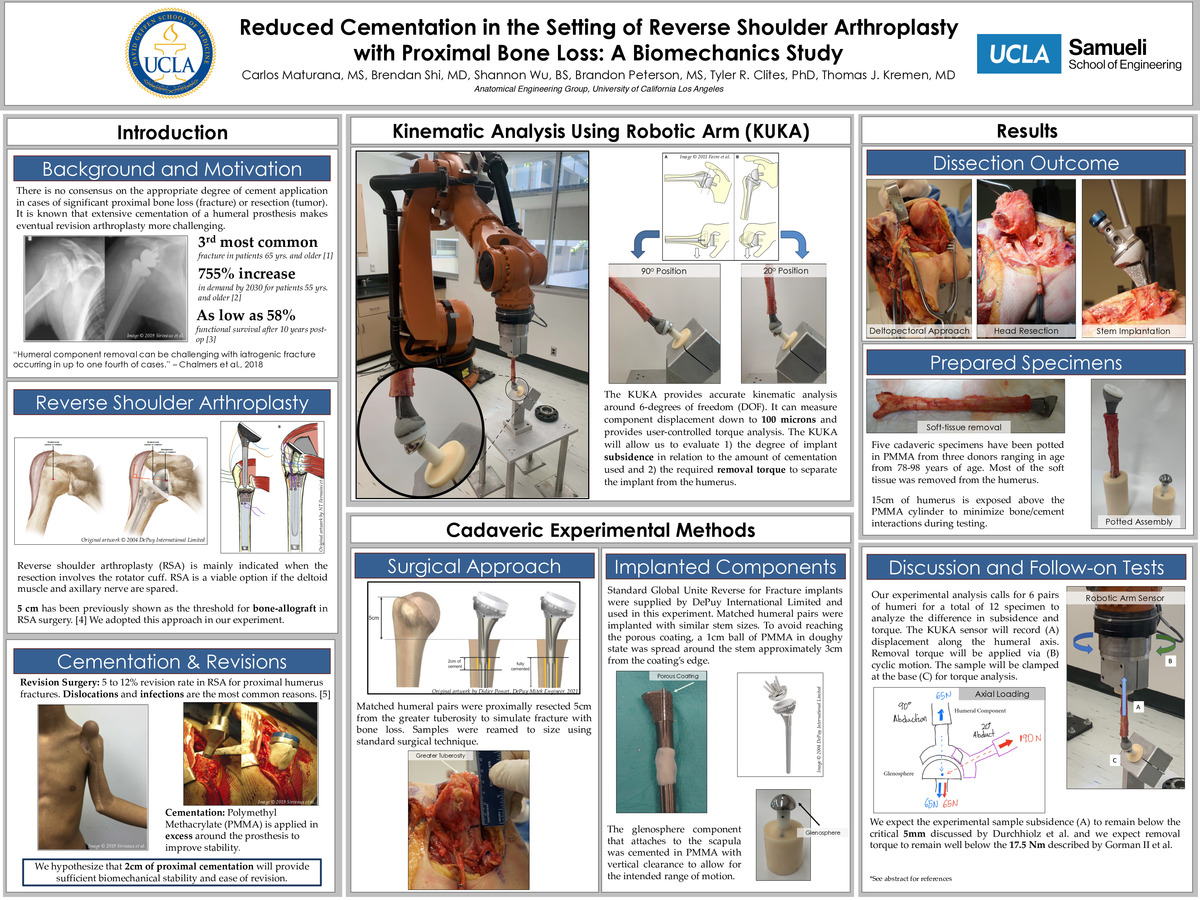
Background: There is a paucity of studies that have evaluated the mechanical benefits of utilizing bone cement (PMMA) during Reverse Shoulder Arthroplasty (RSA). Specifically, there is no consensus on the appropriate degree of cement application in cases of significant proximal bone loss (fracture) or resection (tumor). It is known that extensive cementation of a humeral prosthesis makes eventual revision arthroplasty more challenging and that cemented humeral stems are typically de-bonded from their associated cement mantles via application of a torque force onto the implant. As a result, many surgeons have opted to decrease the degree of cementation at the humeral component in an effort to facilitate less complicated revision surgery in the future. Unfortunately, the exact degree of limited cement application at the humeral component among modern implant designs is unknown. We believe that 2cm of cementation at the proximal smooth portion of the humeral stem will provide sufficient biomechanical stability to resist axial loads, as assessed by less than 5mm of subsidence, while still permitting a reasonably attainable torque threshold for debonding of the cement prosthesis. The aim of this study is to compare the degree of subsidence and torque tolerance between Depuy Global Unite Reverse for fracture prostheses that are fully cemented to the same stem with 2cm proximal cementation.
Materials and Methods: 6 Matched cadaveric humeral pairs [n=12] will be proximally resected 5cm from the greater tuberosity to simulate bone loss due to fracture or tumor. The humeri were divided into (I) 5cm bone loss with stems secured with 2cm proximal cementation and (II) the contralateral humerus with 5cm bone loss with stems fully cemented. Depuy Global Unite Reverse for fracture prostheses were supplied and utilized for all the all experimental setups. After preparation, the humeri and glenosphere component were potted in Polymethyl Methacrylate (PMMA) for use in the kinematic analysis. A 6 degree of freedom (DOF) robotic manipulator is used to perform all testing (KR210; KUKA Robotics Corp., Clinton Township, MI) with the humeral assembly attached to the robot and the glenosphere assembly attached to the base. Synthetic humeri (solid foam, 36cm length and 9.5mm canal diameter [model # 1019]; Synthes USA, Colorado Springs, CO) will be utilized pre-testing to calibrate the robot. The humeral samples will be abducted and adducted from 20 to 90 degrees at 1Hz for 1000 cycles. The assembly will be clamped to the base and subjected to cyclic torque between +/-2.5Nm for 1000 cycles until failure occurs.
Results: 5 cadaveric humeri have been dissected, prepared, and potted in PMMA with the corresponding glenosphere also potted and secured. 4 synthetic Sawbones humeri have been procured and prepared to calibrate The KUKA robot and ensure testing protocols perform as intended. The additional cadaveric specimens will be prepared in the same manner and we are well set to conduct the experiment as described in the methods section.
Discussion and Conclusion: We expect our experimental sample subsidence to remain below the critical 5mm discussed by Durchhiolz et al. as well as the removal torque to remain well below the 17.5 Nm described by Gorman II et al. We expect our hypothesis to hold true and demonstrate that 2cm of cementation provides both sufficient biomechanical stability and ease of removal in cases of revisions.
Level of Evidence: Basic science study.
Reference
1. Kelly BJ, Myeroff CM. Reverse Shoulder Arthroplasty for Proximal Humerus Fracture. Curr Rev Musculoskelet Med. 2020;13(2):186-199. doi:10.1007/s12178-020-09597-0
2. Knowles NK, Columbus MP, Wegmann K, Ferreira LM, Athwal GS. Revision shoulder arthroplasty: a systematic review and comparison of North American vs. European outcomes and complications. J Shoulder Elbow Surg. 2020 May;29(5):1071-1082. doi: 10.1016/j.jse.2019.12.015. Epub 2020 Mar 5. PMID: 32147336.
3. Chalmers PN, Boileau P, Romeo AA, Tashjian RZ. Revision Reverse Shoulder Arthroplasty. J Am Acad Orthop Surg. 2019 Jun 15;27(12):426-436. doi: 10.5435/JAAOS-D-17-00535. PMID: 31170096.
4. Chacon A, Virani N, Shannon R, Levy JC, Pupello D, Frankle M. Revision arthroplasty with use of a reverse shoulder prosthesis-allograft composite. J Bone Joint Surg Am. 2009 Jan;91(1):119-27. doi: 10.2106/JBJS.H.00094. PMID: 19122086.
5. Sirveaux F. Reconstruction techniques after proximal humerus tumour resection. Orthop Traumatol Surg Res. 2019 Feb;105(1S):S153-S164. doi: 10.1016/j.otsr.2018.04.024. Epub 2018 Jun 27. PMID: 29958931.
6. Formaini NT, Everding NG, Levy JC, Rosas S. Tuberosity healing after reverse shoulder arthroplasty for acute proximal humerus fractures: the "black and tan" technique. J Shoulder Elbow Surg. 2015 Nov;24(11):e299-306. doi: 10.1016/j.jse.2015.04.014. Epub 2015 Jul 2. PMID: 26141197.
7. Durchholz, Holger et al. “Core Set of Radiographic Parameters for Shoulder Arthroplasty Monitoring: Criteria Defined by an International Delphi Consensus Process.” JB & JS open access vol. 4,4 e0025. 22 Oct. 2019, doi:10.2106/JBJS.OA.19.00025
8. Gorman RA 2nd, Christmas KN, Simon P, Hess AV 2nd, Brewley EE Jr, Mighell MA, Frankle MA. Optimizing humeral stem fixation in revision reverse shoulder arthroplasty with the cement-within-cement technique. J Shoulder Elbow Surg. 2020 Jul;29(7S):S9-S16. doi: 10.1016/j.jse.2020.01.094. Epub 2020 Apr 29. PMID: 32360178.
9. Favre P, Sussmann PS, Gerber C. The effect of component positioning on intrinsic stability of the reverse shoulder arthroplasty. J Shoulder Elbow Surg. 2010 Jun;19(4):550-6. doi: 10.1016/j.jse.2009.11.044. Epub 2010 Mar 23. PMID: 20335055.
10. Tross AK, Lädermann A, Wittmann T, Schnetzke M, Nolte PC, Collin P, Raiss P. Subsidence of Uncemented Short Stems in Reverse Shoulder Arthroplasty-A Multicenter Study. J Clin Med. 2020 Oct 20;9(10):3362. doi: 10.3390/jcm9103362. PMID: 33092155; PMCID: PMC7590048.
-
PDF
-
Zoom
https://uclahs.zoom.us/j/94927634345?pwd=dHJLTHV1Q2FtOUFjdXNpaHdhY0crQT09