-
Author
Vera Khurshudyan -
PI
Mae Zakhour, MD
-
Co-Author
Averill C, Moatamed N, Memarzadeh S, Zakhour M
-
Title
Mucinous ovarian cancer: a rare entity with variable clinical presentation and management
-
Program
CTSI TL1 Summer Program
-
Other Program (if not listed above)
-
Abstract
Background
Ovarian cancer is the second most common gynecological malignancy, but the most lethal. Mucinous ovarian cancer is a rare form of ovarian cancer that accounts for about 3% of cases. The most common type of ovarian cancer is high-grade serous ovarian cancer accounting for about 65% of cases. Recent molecular analyses have demonstrated that mucinous ovarian adenocarcinomas represent two separate entities, the more indolent expansile subtype and the more aggressive infiltrative subtype. However, these designations are not commonly characterized in pathology reports. In the past treatment for mucinous cancer was solely based on the protocol for serous ovarian cancer. However recent findings have shown that mucinous ovarian cancer is a unique disease that requires its own unique treatment strategy. [1] Early-stage MOC carries an excellent prognosis, with advanced disease having a poor outcome. Surgical management plays an essential role in the early stage and in metastatic disease. Chemotherapy is usually administered for stage II MOC and beyond. The standard gynecology protocol is frequently used, but gastrointestinal regimens have also been administered. [2]
Objectives
- To describe the histologic subtypes and clinical behavior of mucinous ovarian borderline tumors and adenocarcinomas diagnosed at a single institution.
- To describe the treatment strategies used to manage mucinous ovarian cancers over this time period.
Methods
The project is a retrospective chart review of all patients with a diagnosis of mucinous ovarian borderline tumor or adenocarcinoma, diagnosed between January 2010 and December 2020 at Ronald Regan Hospital. The charts were analyzed for several data points including: date of diagnosis, age at diagnosis, smoking history, ethnicity, stage at diagnosis, mucinous histology subtype, size of tumor, primary treatment, type of treatment, date of completion of upfront treatment, recurrence, disease free interval, and current status. The sample size was 44 patients whose diagnosis matched the criteria. Exclusion criteria included neoplasms of non-gynecologic origin. The primary outcomes were to evaluate for consistency in treatment modality determine histologic predictors of clinical outcomes. For statistical analysis a chi-square univariate analysis, unpaired t-test was performed.
Limitations
The main limitation faced was the sample size available at Ronald Regan as mucinous ovarian cancer is not very common. There were 46 patients total which also included those cases with GI cancer that metastasized to the ovaries.
Results
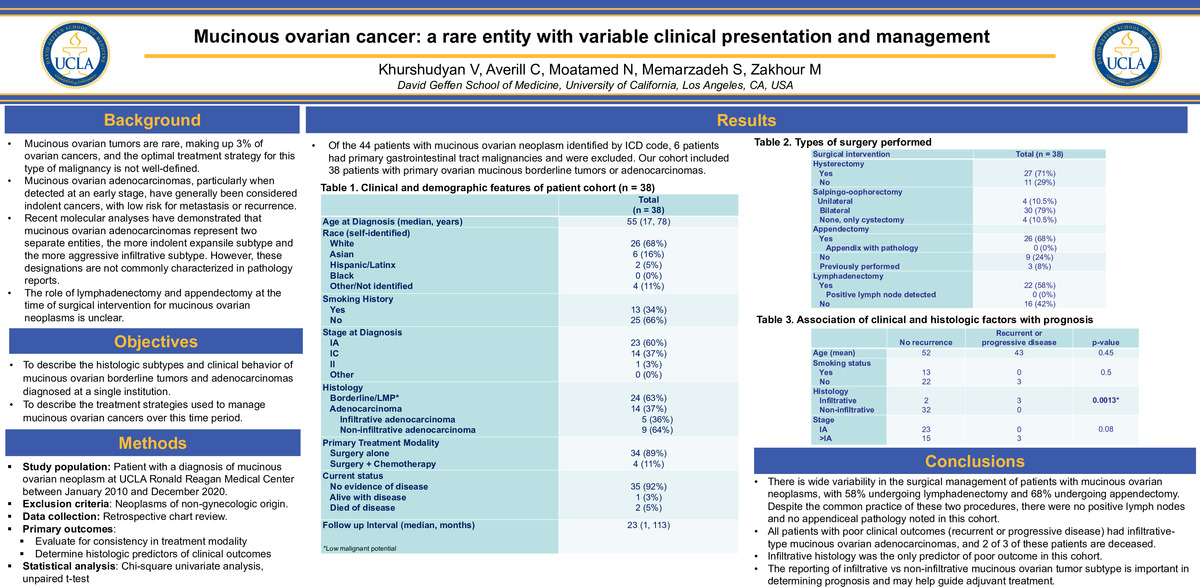
Of the 44 patients with mucinous ovarian neoplasm identified by ICD code, 6 patients had primary gastrointestinal tract malignancies and were excluded. Our cohort included 38 patients with primary ovarian mucinous borderline tumors or adenocarcinomas. The most frequent primary sites that metastasize to the ovary are: 45% from the gastrointestinal tract, 20% from the pancreas, 18% from the cervix and endometrium, and 8% from the breast. From the patients’ charts observed 6 out of 44 patients had gastrointestinal cancer that metastasized to the ovary.
The median age of diagnosis was 55 with a range of 17-78. The highest prevalence as far as race was white accounting for 68%. 66% of the data population had no smoking history. Out of the 38 patients 60% were diagnosed at stage IA, 37% at stage IC, and 3% at stage II. Of those cases, the histology revealed 63% borderline and 37% adenocarcinoma. The primary treatment modality was surgery alone which accounted for 89% of the cases. Currently 92% of the patients have no evidence of disease with a median follow up interval of 23 months.
Conclusion
There is wide variability in the surgical management of patients with mucinous ovarian neoplasms, with 58% undergoing lymphadenectomy and 68% undergoing appendectomy. Despite the common practice of these two procedures, there were no positive lymph nodes and no appendiceal pathology noted in this cohort. All patients with poor clinical outcomes (recurrent or progressive disease) had infiltrative-type mucinous ovarian adenocarcinomas, and 2 of 3 of these patients are deceased. Infiltrative histology was the only predictor of poor outcome in this cohort. The reporting of infiltrative vs non-infiltrative mucinous ovarian tumor subtype is important in determining prognosis and may help guide adjuvant treatment.
According to Morice et al., more than 95% of women with expansile mucinous ovarian cancers present with stage I disease. Cases of the expansile subtype with peritoneal spread are very scarce (only 3 reported cases). In contrast, infiltrative mucinous ovarian cancer is more aggressive, with at least 26% of women presenting with more advanced, non-localized disease at diagnosis; in 17 to 30% of patients who appear to have stage I disease, lymph-node metastases are detected (as compared with no women with expansile mucinous ovarian cancer). Even if the cancer is diagnosed at an early stage, the prognosis for women with infiltrative mucinous ovarian cancer is much poorer, with fatal relapses reported for 15 to 30% of patients with stage I disease. This same outcome was seen in the 38 patients at Ronald Reagan. Thus, the distinction between stage I expansile and stage I infiltrative subtypes is crucial, since it may influence indications for staging lymphadenectomy or adjuvant chemotherapy. [1]
In the future, we are hoping to also have pathology look at the cancer samples and run staining tests for mutational landscape. The first thing we would like to test is the KRAS gene as mucinous ovarian cancer is known to be KRAS driven. Next, we may analyze the HER2 and P53 genes that were noted to be common in the infiltrative subtype.
Through these different reviews we hope to discover a better method of treatment for the specialized subtypes of mucinous ovarian cancer. Currently there is no specialized treatment specifically for this type of cancer therefore there may be overtreatment or under treatment of patients. If mutational markers are discovered this may lead to more genetic testing of those with family history of mucinous ovarian cancer. Once a correlation is determined we hope to spread this data and possibly start a clinical trial for the treatment processes. Currently there are no active clinical trials specifically for mucinous ovarian cancers.
References
[1] Morice P, Gouy S, Leary A. Mucinous Ovarian Carcinoma. N Engl J Med. 2019 Mar 28;380(13):1256-1266. doi: 10.1056/NEJMra1813254. PMID: 30917260.
[2] Babaier A, Ghatage P. Mucinous Cancer of the Ovary: Overview and Current Status. Diagnostics (Basel). 2020 Jan 19;10(1):52. doi: 10.3390/diagnostics10010052. PMID: 31963927; PMCID: PMC7168201.
-
PDF
-
Zoom
https://uclahs.zoom.us/j/97810150855?pwd=SGtkbE11VSswN2RuOXVWL1ArUXhaUT09