-
Author
Rogelio Medina -
Co-author
Rogelio Medina, BA
-
Title
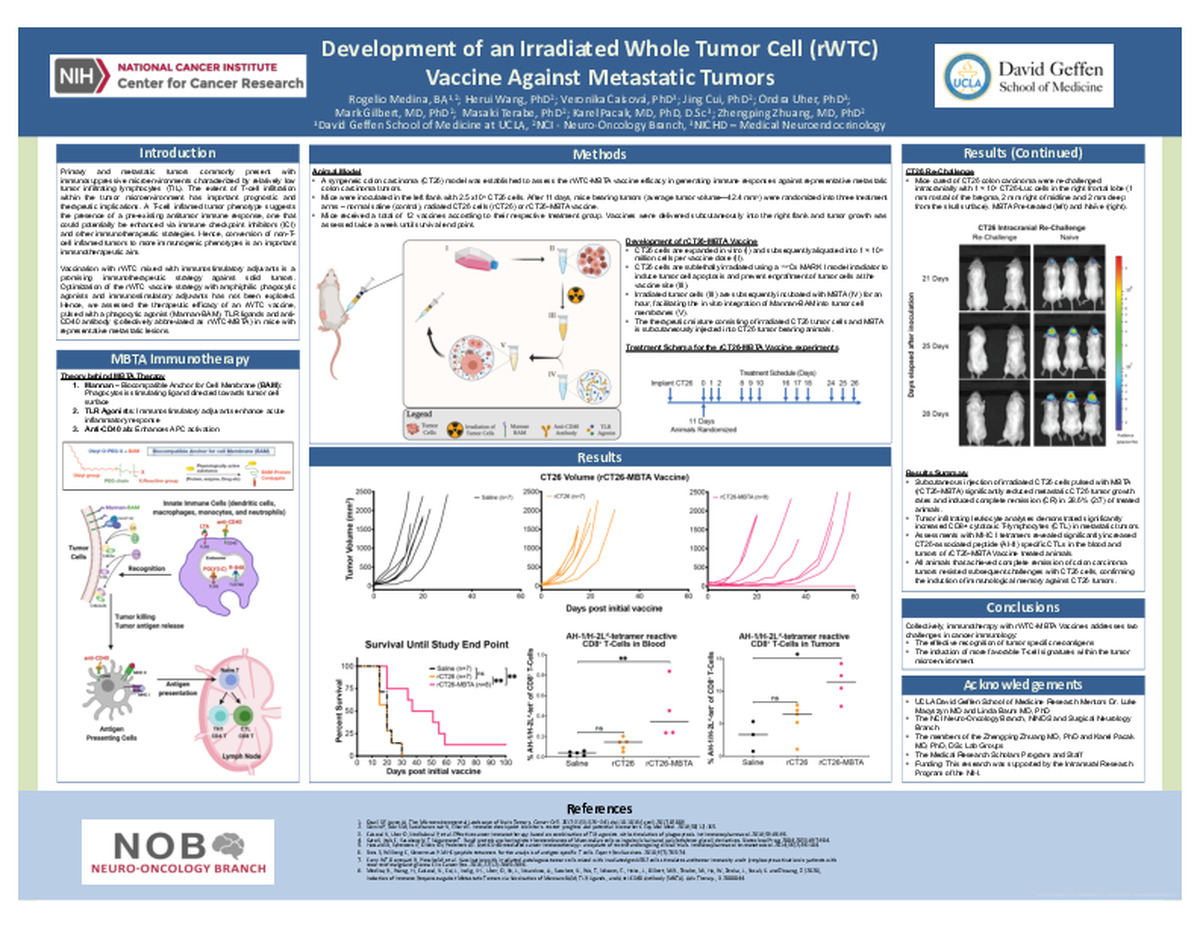
Development of an Irradiated Whole Tumor Cell (rWTC) Vaccine Against Metastatic Tumors
-
Abstract
Introduction: Emerging evidence is demonstrating the extent of T-cell infiltration within the tumor microenvironment has favorable prognostic and therapeutic implications. Hence, immunotherapeutic strategies that augment the T-cell signature of tumors hold promising therapeutic potential. Here, we assessed the therapeutic efficacy of an irradiated whole tumor cell (rWTC) vaccine, pulsed with a phagocytic agonist (Mannan-BAM), TLR ligands and anti-CD40 antibody (collectively abbreviated as rWTC-MBTA) to control tumor growth and improve survival in mice with metastatic tumors.
Methods: A syngeneic colon carcinoma (CT26) model was established to assess the rWTC-MBTA vaccine efficacy in generating immune responses against representative metastatic colon carcinoma tumors.
Results: Subcutaneous injection of irradiated CT26 cells pulsed with MBTA (rCT26-MBTA) significantly reduced metastatic CT26 tumor growth rates and induced complete remission (CR) in 28.6% (2/7) of treated animals. Tumor infiltrating leukocyte analyses demonstrated significantly increased CD8+ cytotoxic T-lymphocytes (CTL) in metastatic tumors. Further assessments with MHC I tetramers revealed significantly increased CT26-associated peptide (AH1) specific CTLs in the blood and tumors of rCT26-MBTA Vaccine treated animals. All animals that achieved complete remission of colon carcinoma tumors resisted subsequent challenges with CT26 cells, confirming the induction of immunological memory against CT26 tumors.
Conclusion: Our investigation demonstrates that rWTC-MBTA Vaccines can effectively induce a tumor-specific adaptive immune response that can target metastatic tumors.
-
College
AAC
-
Zoom
-
PDF